Successfully Added to Cart
Customers also bought...
-
 DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the...
DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the... -
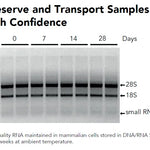
 DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
Format
ZymoBIOMICS Microbial Community DNA Standard
Highlights
- Accurate composition: composition cross-validated with multiple types of measurements.
- Negligible impurity: guaranteed to contain < 0.01% foreign microbial DNA.
- Wide range of GC content: 15%-85%, for assessing bias caused by GC content variation.
Original Manufacturer
Satisfaction 100% guaranteed, read Our Promise
Innovated in California, Made in the USA
Format
ZymoBIOMICS Microbial Community DNA Standard
Highlights
- Accurate composition: composition cross-validated with multiple types of measurements.
- Negligible impurity: guaranteed to contain < 0.01% foreign microbial DNA.
- Wide range of GC content: 15%-85%, for assessing bias caused by GC content variation.
Original Manufacturer
Satisfaction 100% guaranteed, read Our Promise
Innovated in California, Made in the USA
| Cat # | Name | Size | Price | Quantity |
|---|
Description
Performance
Technical Specifications
| Purity | < 0.01% foreign microbial DNA |
|---|---|
| Sample Source | A mixture of genomic DNA of ten microbial strains. |
| Sample Storage | -20 °C |
Resources
Documents
FAQ
We recommend working backwards from the analysis. First optimize your library prep with the ZymoBIOMICS Microbial Community DNA Standard (D6305, D6311) to assess bias in PCR, sequencing, bioinformatics, etc. Once your results have low/no bias when compared to the theoretical composition, then use the whole cell ZymoBIOMICS Microbial Community Standard (D6300, D6310) to assess bias in lysis efficiency.
No, the Microbial Community Standard is designed to assess the efficiency of the lysis method and is intended to be run in parallel with your samples. If all the organisms can be detected at/near the theoretical abundance, you can be confident your extraction method is unbiased.
The chemistry of the Qiagen Kits (QIAamp Powerfecal, DNeasy Powersoil) and the storage solution the Microbial Community Standard is stored in (DNA/RNA Shield) are not completely compatible. Instead, less input volume should be used (25 ul).
You can find the reference genome and 16S/18S sequences here: ZymoBIOMICS.STD.refseq.v2.zip.
This may indicate an issue with the lysis method, which can be biased toward gram negative bacteria. See the Optimized Lysis Protocols under Documents for a table of bead beating devices and protocols validated by Zymo Research.
Devices found to underrepresent tough-to-lyse organisms under all tested conditions:
- Retsch Mixer Mills
- Tissues Lyzers
- MP Fast Prep 96
We use an in-house curated database to generate the data for the standards.
Please contact Technical Support at tech@zymoresearch.com for raw sequencing data.
The expected DNA fragment size is <15kb.
This could indicate bias in the workflow, such as inefficient lysis, library prep or bioinformatics analysis.
Citations
Workflow Generator
Build Your Custom Microbiome Workflow
A great tool to help build or complete your workflow with our comprehensive microbiome solutions!
Need help? Contact Us




