Successfully Added to Cart
Customers also bought...
-
 DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the...
DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the... -
 DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
Highlights
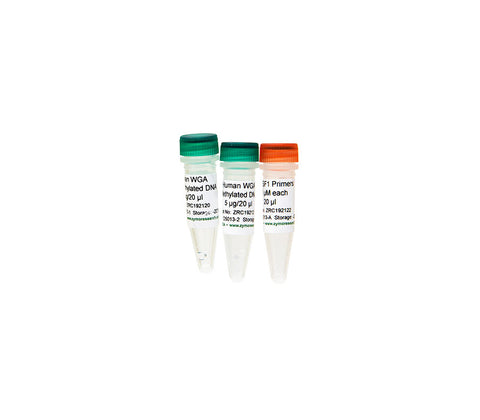
- Purified, non-methylated and methylated human DNA for use as negative and positive control in methylation detection applications.
- Standards can be assayed in parallel with samples to monitor bisulfite conversion efficiency.
- Ideal controls for bisulfite sequencing PCR (BSP) and methylation specific PCR (MSP). Primers included with the full set are designed to amplify non-methylated, methylated, and mixed methylated copies of the Ras association (RalGDS/AF-6) domain family member 1 (RASSF1) gene following bisulfite conversion.
Original Manufacturer
Satisfaction 100% guaranteed, read Our Promise
Innovated in California, Made in the USA
Highlights
- Purified, non-methylated and methylated human DNA for use as negative and positive control in methylation detection applications.
- Standards can be assayed in parallel with samples to monitor bisulfite conversion efficiency.
- Ideal controls for bisulfite sequencing PCR (BSP) and methylation specific PCR (MSP). Primers included with the full set are designed to amplify non-methylated, methylated, and mixed methylated copies of the Ras association (RalGDS/AF-6) domain family member 1 (RASSF1) gene following bisulfite conversion.
Original Manufacturer
Satisfaction 100% guaranteed, read Our Promise
Innovated in California, Made in the USA
Description
Performance
Technical Specifications
| Concentration | 250 ng/µl in buffer (10 mM Tris-HCl, 1 mM EDTA, pH 8.0) |
|---|---|
| Sample Source | Whole genome amplified DNA from HCT116 DKO cells. Human WGA Methylated DNA is enzymatically methylated by M.SssI methyltransferase. |
| Sample Storage | ≤ -20°C |
Resources
Documents
FAQ
All standards are enzymatically methylated by CpG Methylase in vitro at all CpG dinucleotides. The methylated DNA is very highly methylated (> 95% of all CpG sites).
D5014 is comprised of genomic DNA isolated from a HCT116 DKO cell line. The D5013 standard is comprised of the same HCT116 DKO DNA but has undergone whole genome amplification (WGA). D5013 may be more suitable for researchers working with WGA experimental samples. As a result of the WGA process, any residual methylation marks are removed from the unmethylated WGA standard (D5013-1), resulting in ultra-low methylation levels.
These standards can be adapted to many assay types. In addition to BSP and MSP, these standards can serve as controls in bisulfite library preparation and methylation arrays. For more detailed information, please visit our page on Controls For DNA Methylation Assays.
In experimental samples with unknown methylation patterns, it may be difficult to determine if cytosines remaining after bisulfite conversion are the result of non-CpG methylation, or inefficient conversion. Because DNA standards are known to have very low non-CpG methylation, these can be run in parallel as controls to assess the efficiency of the conversion reaction.
These standards are included with primers targeting RASSF1, and will amplify non-methylated, methylated, and mixed methylated templates. By amplifying bisulfite converted DNA with the included primers, researchers can test and optimize their bisulfite PCR conditions.
We recommend ZymoTaq DNA Polymerase, a hot start Taq polymerase specifically designed to amplify bisulfite converted DNA. Another hot start DNA polymerase of choice may also be used.
These standards can be used for optimizing bisulfite library preparation, and it can be used as a positive control to ensure the library prep reagents are working optimally. However, if the desire is to assess bisulfite conversion rate, it is best to use E. coli Non-methylated Genomic DNA (Catalog No. D5016) as an in situ control. Contact technical support tech@zymoresearch.com for additional information.
The standards are stable at ≤ -20°C for up to 24 months. For long term storage standards should be stored at ≤ -70°C.
Citations
Need help? Contact Us