Successfully Added to Cart
Customers also bought...
-
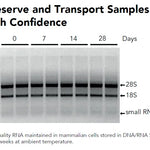 DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the...
DNA/RNA Shield (50 ml)Cat#: R1100-50DNA/RNA Shield reagent is a DNA and RNA stabilization solution for nucleic acids in any biological sample. This DNA and RNA stabilization solution preserves the... -
 DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
DNA/RNA Shield SafeCollect Swab Collection Kit (1 ml fill) (1 collection kit)Cat#: R1160The DNA/RNA Shield SafeCollect Swab Collection Kit is a user-friendly collection kit for stabilizing the nucleic acid content of samples collected with a swab. DNA/RNA...
Highlights
- Fast: Yeast cells with high transformation efficiencies can be prepared in under 10 minutes.
- Simple: Easy method to transform with single or multiple plasmids in ≤ 1 hour with carrier DNA.
- Versatile: Can be used with S. cerevisiae, as well as other fungi, including C. albicans, S. pombe, and P. pastoris. Compatible with both circular and linear DNA.
Original Manufacturer
100% satisfaction guaranteed, read Our Promise
Innovated in California, Made in the USA
Highlights
- Fast: Yeast cells with high transformation efficiencies can be prepared in under 10 minutes.
- Simple: Easy method to transform with single or multiple plasmids in ≤ 1 hour with carrier DNA.
- Versatile: Can be used with S. cerevisiae, as well as other fungi, including C. albicans, S. pombe, and P. pastoris. Compatible with both circular and linear DNA.
Original Manufacturer
100% satisfaction guaranteed, read Our Promise
Innovated in California, Made in the USA
| Cat # | Name | Size | Price | |
|---|---|---|---|---|
| T2002 | Frozen-EZ Yeast Solution I | 60 ml | €35,70 | |
| T2003 | Frozen-EZ Yeast Solution II | 6 ml | €47,90 | |
| T2004 | Frozen-EZ Yeast Solution III | 60 ml | €64,20 | |
Description
Performance
Technical Specifications
| Competent Cell Stability | ≥ 1 year at -70°C |
|---|---|
| Processing Time | ≤ 15 minutes |
| Product Storage | 0 - 4°C |
| Transformation DNA Input | 0.2 - 1.0 µg |
| Transformation Efficiency | 105 - 106 cfu/µg plasmid DNA. |
Resources
Documents
FAQ
The procedure utilized in this kit is designed, in some ways, similar to the lithium cation based method. No spheroplast step is involved. The mechanism probably involves some metabolic pathways that we do not fully understand.
Yes. Based on data from other labs and ours, this kit does work as well on C. albicans, S. pombe, or Pichia pastoris as well as S. cerevisiae.
There is no loss of transformation efficiency after half a year of storage below -70°C. The transformation efficiency gradually starts to drop after 6 months.
Thaw at room temperature.
We usually see a 10-30% increase of transformants after the first cycle of freezing. You can refreeze and thaw the competent yeast cells 3-4 times without noticeable effect on the transformation efficiency. Further freezing-thawing cycles adversely affect the transformation efficiency.
No. Temperatures between 30°C and 37°C are in the optimal temperature range. Incubation below or above this range greatly reduces the transformation efficiency.
Yes. Different digestion buffers have only a slight effect on the transformation efficiency. You should try to keep the DNA volume in 5 µl per transformation experiment by increasing the concentration of DNA in the digestion reaction.
For most circular plasmid transformations, 50 µl is enough. But if you use linearized DNA or use more than one selection marker, you can apply up to 200 µl of transformation mixture on each plate to increase the number of transformants. The number of transformants increases linearly with the amount of transformation mixture applied to each plate.
No. Use any plate that is appropriate for your experiment.
No. There is no need for carrier DNA.
Citations
Need help? Contact Us




